1. Warning
The active ingredient of this drug is Botulinum Toxin Type A, produced by Clostridium botulinum. Therefore, it is crucial to be aware of and adhere strictly to the prescribed usage and dosage. Physicians administering this drug must have a thorough understanding of the anatomical structure of the related neuromuscular and periorbital areas, any anatomical alterations due to previous surgeries, and standard electromyographic techniques.
The recommended dosage and frequency of administration must not be exceeded.
A. Distant Spread of Toxin Effect
Botulinum toxin can spread from the site of injection to other areas, potentially causing botulism. Symptoms such as rapid muscle weakness, fatigue, hoarseness, speech disorders, stuttering, loss of bladder control, respiratory difficulty, swallowing difficulty, double vision, blurred vision, and drooping eyelids may occur. Symptoms such as respiratory difficulty or swallowing difficulty can be life-threatening, and there have been reported cases of death due to the spread of the toxin. Children treated for spastic cerebral palsy are particularly at high risk. However, adults treated for spastic cerebral palsy or other conditions may also exhibit the same symptoms. There have been instances of such adverse reactions occurring at the recommended doses for treating cervical dystonia, as well as at lower doses.
B. Hypersensitivity Reactions
Rare cases of serious or immediate hypersensitivity reactions to other botulinum toxin products have been reported. These reactions have included anaphylaxis, urticaria, tissue swelling, and respiratory difficulties. One instance of anaphylaxis was reported when lidocaine was used as a solvent; however, the causative agent was not conclusively identified. If such reactions occur following the administration of this drug, the treatment should be discontinued and appropriate measures taken.
C. In Patients with Neuromuscular Disorders
Patients with peripheral motor neuropathic diseases (e.g. amyotrophic lateral sclerosis, motor neuropathy) or neuromuscular junction disorders (e.g. myasthenia gravis, Lambert-Eaton syndrome) may experience an increased risk of significant systemic reactions, including severe dysphagia and respiratory depression, even with conventional doses of botulinum toxin products. According to clinical literature, rare cases have been reported where patients with known or unrecognized neuromuscular disorders exhibited severe hypersensitivity to the systemic effects of typical doses of botulinum toxin. In some of these cases, dysphagia persisted for several months, necessitating the use of a feeding tube.
D. Dysphagia
Dysphagia is a commonly reported adverse reaction in patients treated with any botulinum toxin for cervical dystonia. Rarely, dysphagia has been severe enough in some patients to require the use of a feeding tube. There have been reports of cases where dysphagia led to aspiration pneumonia, resulting in death.
E. Rarely, cardiovascular adverse reactions, including arrhythmias and myocardial infarctions, have been reported following the administration of other botulinum toxin products, some of which have been fatal. Several of these cases involved patients with pre-existing cardiovascular risk factors.
F. In the treatment of strabismus with other botulinum toxin products, retrobulbar hemorrhage, which can jeopardize retinal circulation, has occurred due to needle penetration around the eye. It is recommended to use appropriate techniques to reduce the pressure susceptible around the eye. There have also been cases of needle penetration into the eye. Ophthalmoscopic examination should be used to diagnose these conditions. One or more external ocular muscle paralyses can induce spatial disorientation, double vision, and past-pointing. Covering the affected eye can alleviate these symptoms.
G. Eyelid Spasm
After the injection of botulinum toxin into the orbicularis oculi muscle, especially in patients with facial nerve (CN VII) disorders, there can be a reduction in blinking leading to corneal exposure, persistent epithelial defects, and corneal ulcers. There has been a reported case where corneal perforation occurred in an anophthalmic eye due to the effects of another botulinum toxin product, necessitating a corneal transplant. In patients with a history of eye surgery, a careful examination of corneal sensation should be conducted, and avoiding injections in the lower eyelid area can reduce the risk of ectropion. Any corneal epithelial defects require thorough treatment. This may include protective eye drops, ointments, therapeutic soft lenses, or eye patches, among other measures.
H. Non-Interchangeability
As the toxin content can vary between different botulinum toxin products, the units of one product are not convertible to the units of another.
I. Injection into or around Anatomically Vulnerable Structures
Caution is required when injecting into or around anatomically vulnerable structures. Serious adverse events, including fatal outcomes, have been reported with the injection of other botulinum toxin products into the salivary glands, the area around the mouth-tongue-pharynx, esophagus, and stomach. Some of these patients had pre-existing dysphagia or significant weakness. (The safety and efficacy for these indications, including those injection sites, have not been established.) Cases of pneumothorax associated with the injection process of other botulinum toxin products into the thoracic region have been reported. Caution is especially advised when injecting near the apex of the lung.
In patients with respiratory disorders undergoing treatment for upper limb spasticity, a decrease in pulmonary function (a reduction in Forced Vital Capacity [FVC] by ≥ 15%) and upper respiratory infections have been reported more frequently with the administration of other botulinum toxin products compared to placebo. Also, a decrease in pulmonary function (FVC reduction ≥ 15%) has been reported in patients with respiratory disorders and neurogenic detrusor overactivity treated with other botulinum toxin products.
K. Bronchitis and Upper Respiratory Infections in Adult Patients Treated for Upper Limb Spasticity
Among patients treated for upper limb spasticity, bronchitis has been reported more frequently in those treated with other botulinum toxin products than in the placebo group. In patients with reduced lung function undergoing treatment for upper limb spasticity, upper respiratory infections have been reported more frequently with other botulinum toxin products compared to placebo. In a therapeutic confirmatory clinical trial conducted on 195 adults for the treatment of upper limb spasticity using this drug, there were two cases of upper respiratory infection adverse reactions reported in the treatment group and one case in the control group.
2. Do Not Administer to the Following Patients:
A. Patients who have hypersensitivity to the components of this drug
B. Patients with systemic neuromuscular junction disorders (such as myasthenia gravis, Lambert-Eaton syndrome, amyotrophic lateral sclerosis, etc.) - The muscle-relaxing action of the drug can exacerbate these conditions.
C. Pregnant or potentially pregnant women and nursing mothers
3. Administer with Caution to the Following Patients
A. Patients currently being treated with muscle relaxants (such as tubocurarine chloride, dantrolene sodium, etc.) - There is a risk of enhanced muscle-relaxing action or increased incidence of dysphagia.
B. Patients receiving drugs with muscle-relaxing properties, such as spectinomycin hydrochloride, aminoglycoside antibiotics (gentamicin sulfate, neomycin sulfate, etc.), polypeptide antibiotics (polymyxin B sulfate, etc.), tetracycline antibiotics, lincosamide antibiotics (lincosamides), muscle relaxants (baclofen, etc.), anticholinergics (scopolamine butylbromide, trihexyphenidyl hydrochloride, etc.), benzodiazepines and similar drugs (diazepam, etizolam, etc.), and benzamide drugs (tiapride hydrochloride, sulpiride, etc.) - There is a risk of enhanced muscle-relaxing action or increased incidence of dysphagia.
4. Adverse Reactions
A. General Information
Rare cases of difficulty swallowing, pneumonia, and/or severe lethargy or anaphylaxis-related deaths have been reported following treatment with botulinum toxin.
Additionally, cardiovascular adverse reactions, including arrhythmia or myocardial infarction that can lead to fatal outcomes, have been rarely reported. The precise causal relationship between these adverse reactions and botulinum toxin has not been established.
Other botulinum toxin products have reported adverse reactions, whose association with botulinum toxin remained unknown, including skin rashes (multiform erythema, urticaria, psoriatic rash), itching, and allergic reactions.
Adverse reactions typically appear within a week of injection, are usually transient but can persist for several months. Local pain, tenderness, bruising, tightness at the injection site, swelling, warmth at the injection site, and increased muscle tension in the injection area and neighboring muscles can occur in relation to the injection. Local weakness at the injection site reflects the anticipated pharmacological action of botulinum toxin. However, weakness in adjacent muscles may be attributed to the spread of the toxin. In patients treated with this drug for eyelid spasms or cervical muscle dystonia, there is a possibility of increased electromyographic jitter (sudden disruption in waveform) unrelated to electromyographic abnormalities in muscles distant from the injection site.
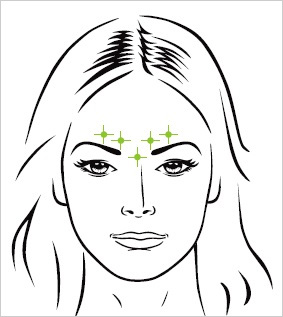
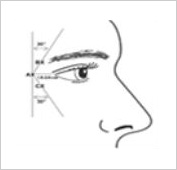
B. Glabellar Lines
A safety assessment was conducted on 268 subjects of ages of ages over 20 to under 65 with moderate to severe glabellar lines across multiple centers. This was a double-blind (neither patients nor doctors were aware), randomized, active-controlled study (135 subjects in the test group for this drug, 133 in the Botox control group). Adverse events occurred in 20.00% of the test group and 18.05% of the control group.
Most adverse events were mild, with no severe cases reported. The drug-related adverse reactions reported in more than 1% of the test group for this drug, in order of frequency, were as follows: Eyelid ptosis (2.22%), eyebrow elevation (1.48%), and dizziness (1.48%).
A safety assessment was conducted on 197 subjects of ages 18 and above who were diagnosed with a stroke at least 6 weeks prior. This was a multi-center, active-controlled, double-blind (where neither patients nor doctors are aware of the study), randomized study (99 subjects in the test group for this drug, 98 in the Botox control group). Adverse events were reported in 19.59% (19 out of 97 subjects, 30 events) of the test group and 19.39% (19 out of 98 subjects, 22 events) of the control group. Among these, drug-related adverse reactions were reported in 3 cases in the test group and 4 cases in the control group. The table below lists the reported drug-related adverse reactions by body system. The frequencies are defined as follows: very common (≥ 10%), common (≥ 1%, < 10%), uncommon (≥ 0.1%, < 1%), rare (≥ 0.01%, < 0.1%), very rare (< 0.01%).
| Table 1. Drug-Related Adverse Reactions Associated with Treatment in the Test Group for This Drug |
| Body System |
Common(≥1%, <10%) |
Musculoskeletal and
Connective Tissue Disorders |
Muscle Weakness (1.03%, 1 case)
Limb Pain (1.03%, 1 case)
Muscle Atrophy (1.03%, 1 case)
|
D. Lateral Canthal Lines (Crow's Feet)
In a multicenter study involving 204 participants aged between 18 and 75 years with moderate to severe lateral canthal lines, a safety assessment was conducted using an active control, double-blind (a clinical trial method where neither the patients nor doctors know who is receiving treatment), and randomized assignment. Adverse reactions unrelated to the lateral canthal line area were observed in 9.80% of the subjects (20 out of 204 subjects, 28 events). There were 9 mild cases and 19 moderate cases. None were acute reactions, and they were not related to the treatment with this drug.
Drug-related adverse reactions in the area of lateral canthal lines occurred in 1 subject (0.49%, 1 event each) in both the test and control groups, and all were mild in severity. (Test group administration site: Itching , Control group administration site: Tenderness)
E. Eyelid Spasm
A safety assessment was conducted on 230 patients diagnosed with essential eyelid spasms, aged 18 and above, in a multicenter study using active control, double-blind (neither patients nor doctors were aware), and randomized assignment (test group of this drug: 117 subjects, Botox control group: 113 subjects). Adverse events occurred in 39.32% of the test group (46 out of 117 subjects, 76 events) and 27.43% of the control group (31 out of 113 subjects, 51 events). Among these, drug-related adverse reactions associated with the treatment occurred in 11.97% of the test group (14 out of 117 subjects, 16 events) and 13.27% of the control group (15 out of 113 subjects, 18 events). The table below lists the reported drug-related adverse reactions by body system.
<Table 2>
| Body System |
Test Group (117 Subjects) |
Control Group (113 Subjects) |
| General Disorders and Administration Site Conditions |
|
|
| Bruising at Injection Site |
6 subjects (5.13%, 6 events) |
5 subjects (4.42%, 5 events) |
| Swelling at Injection Site |
2 subjects (1.71%, 2 events) |
5 subjects (4.42%, 5 events) |
| Inflammation at Injection Site |
1 subject (0.85%, 1 event) |
0 subjects (0.00%, 0 events) |
| Eye Disorders |
|
|
| Eyelid Inflammation |
1 subject (0.85%, 2 events) |
1 subject (0.88%, 1 event) |
| Increased Tear Production |
0 subjects (0.00%, 0 events) |
2 subjects (1.77%, 2 events) |
| Dry Eye |
1 subject (0.85%, 1 event) |
0 subjects (0.00%, 0 events) |
| Eyelid Drooping |
1 subject (0.85%, 1 event) |
0 subjects (0.00%, 0 events) |
| Itching Eye |
0 subjects (0.00%, 0 events) |
1 subject (0.88%, 1 event) |
| Foreign Body Sensation in Eyes |
1 subject (0.85%, 1 event) |
0 subjects (0.00%, 0 events) |
| Abnormal Lacrimation |
1 subject (0.85%, 1 event) |
0 subjects (0.00%, 0 events) |
| Rabbit Eye Syndrome |
0 subjects (0.00%, 0 events) |
1 subject (0.88%, 1 event) |
| Skin and Subcutaneous Tissue Disorders |
|
|
| Itching |
0 subjects (0.00%, 0 events) |
2 subjects (1.77%, 2 events) |
| Nervous System Disorders |
|
|
| Dizziness |
1 subject (0.85%, 1 event) |
0 subjects (0.00%, 0 events) |
| Respiratory Disorders |
|
|
| Breathing Difficulty |
0 subjects (0.00%, 0 events) |
1 subject (0.88%, 1 event) |
F. Post-Market Surveillance Results in South Korea
□ Glabellar Lines
Over 4 years, a post-market usage survey was conducted in South Korea with 914 subjects. The incidence of adverse events was 2.5% (23 out of 914 subjects, 25 events). Of these, the incidence of drug adverse reactions with a possible causal relationship to this product was 1.6% (15 out of 914 subjects, 17 events). Reported adverse events included headaches at 0.5% (5 out of 914 subjects, 5 events), eye fatigue and periorbital edema each at 0.2% (2 out of 914 subjects, 2 events), and chills, fatigue, facial paralysis, reaction at injection site, rash at injection site, pressure sensation at injection site, pain at injection site, and itching each at 0.1% (1 out of 914 subjects, 1 event). No serious adverse events or serious drug adverse reactions were identified.
The incidence of unexpected adverse events was 1.5% (14 out of 914 subjects, 16 events), with headaches at 0.8% (7 out of 914 subjects, 7 events), eye fatigue and pharyngitis each at 0.2% (2 out of 914 subjects, 2 events), and back pain, chills, fatigue, facial paralysis, and skin wrinkling each at 0.1% (1 out of 914 subjects, 1 event). Among these, the incidence of unexpected drug adverse reactions that cannot be ruled out as causally related to this product was 0.9% (8 out of 914 subjects, 10 events), with headaches reported at 0.5% (5 out of 914 subjects, 5 events), eye fatigue at 0.2% (2 out of 914 subjects, 2 events), and chills, fatigue, facial paralysis each reported at 0.1% (1 out of 914 subjects, 1 event). No serious and unexpected adverse events or serious and unexpected drug adverse reactions were identified.
Upon the comprehensive evaluation at the time of re-examination conclusion, using the domestic post-marketing adverse event and spontaneous adverse reaction report data for this drug in comparison with the reported adverse event data for all other approved drugs in South Korea, the following newly identified adverse events were reported more statistically significantly in this drug compared to other drugs. However, these results do not imply a proven causal relationship between the respective ingredient and the following adverse events:
• Skin and Subcutaneous Tissue Disorders: Periorbital Edema (Eyelid Edema)
• Nervous System Disorders: Headache
5. General Precautions
A. This drug contains human albumin derived from human blood. When administering drug products manufactured from human blood or plasma, the possibility of transmission of infectious agents cannot be completely excluded. This also applies to pathogens that are currently unknown. To reduce the risk of transmission of infectious agents, the manufacturing process includes appropriate measures such as the screening of donors and donation sites, as well as processes for the removal and/or inactivation of infectious agents.
B. The impact of this drug on the ability to operate machinery or drive cannot be predicted, as it may be influenced by the disease being treated.
C. Glabellar Lines/Lateral Canthal Lines (Crow's Feet)
The reduction in blinking following the injection of botulinum toxin products into the muscles around the eyes can lead to corneal exposure, persistent epithelial defects, and corneal ulceration, especially in patients with 7th nerve disorders.
Caution is advised in cases such as when there is infection or skin diseases, scars, or other skin abnormalities at the injection site, a history of facial plastic surgery (face lifting), treatment with permanent implants, a history of facial nerve paralysis or eyelid spasms, or when physical improvement of glabellar lines is unlikely, such as when wrinkles do not smooth out upon manually spreading them. These patients were excluded from Phase 3 safety and efficacy clinical trials.
The interval between injections of this drug should not be more frequent than every three months, and the minimum effective dose should be used.
D. Upper Limb Spasticity
This drug is only used for the treatment of focal spasticity studied in conjunction with conventional standard therapies. It is not effective in improving the range of motion in joints affected by fixed contractures.
6. Drug Interactions
A. Generally, when botulinum toxin products are taken in conjunction with aminoglycoside antibiotics or drugs that interfere with muscle and nerve transmission (such as tubocurarine muscle relaxants), the efficacy of the botulinum toxin products is increased. Continuous concurrent use of aminoglycoside antibiotics or spectinomycin is contraindicated.
The use of polymyxin, tetracycline, or lincomycin in patients treated with this drug should be approached with caution.
B. The effects of administering a different botulinum neurotoxin serotype concurrently or within several months are not known. Administering a different botulinum toxin before the effects of a previously administered botulinum toxin have dissipated may excessively exacerbate muscle and nerve weakness.
7. Administration in Pregnant and Nursing Women
There are no adequate and well-controlled studies of this drug in pregnant women. In studies where pregnant rats were injected intramuscularly with this drug (0.5, 1, 4 U/kg) during the organogenesis period (day 6-16 of pregnancy), maternal effects such as bent toes, paralytic gait, and curled toes were observed. In the fetuses, a decrease in ossification numbers was noted in the 4 U/kg dosage group, but this was not statistically significant.
It is not known whether botulinum toxin is secreted in human milk. Since many drugs are excreted in human milk, caution should be exercised when administering the drug to nursing mothers.
The use of this drug is not recommended during pregnancy or breastfeeding.
8. Administration in Children
This drug has not been studied for safety and efficacy in children and adolescents under 20 years of age for the improvement of glabellar lines.
The safety and efficacy for upper limb spasticity have not been studied in children and adolescents under 18 years of age.
9. Carcinogenicity, Mutagenicity, Teratogenicity, Animal Toxicity
Long-term studies to evaluate the carcinogenic potential in animals have not been conducted.
10. Treatment for Overdose
Symptoms and signs of overdose may not be immediately apparent post-injection.
In cases of accidental administration or oral ingestion, medical observation for signs or symptoms of systemic weakness or muscle paralysis should be conducted for several weeks. If an overdose or misadministration is immediately recognized, an antitoxin can be used. The antitoxin does not reverse muscle weakness effects that have already manifested at the time of its administration.
If the muscles of the oropharynx and esophagus are affected, aspiration may occur, which can lead to aspiration pneumonia. If respiratory muscles are paralyzed or weakened, intubation and assisted breathing may be necessary until recovery. In addition to other general supportive treatments, tracheotomy and/or prolonged mechanical ventilation may be required.
Such patients should consider immediate and appropriate measures, including additional medical evaluation and hospitalization.
11. Storage and Handling Precautions
Unopened drug must be stored refrigerated (2-8℃). Once reconstituted, the drug can be refrigerated (2-8℃) for up to 24 hours.
All vials, including those past their expiration date, and any supplies that have come into direct contact with the product, must be disposed of as medical waste. In cases requiring deactivation of the toxin (e.g. spillage), it is recommended to use diluted hypochlorite (0.5 or 1%) before disposal as medical waste.
12. Information for Patients
Discuss any concerns regarding the effects and risks of this drug with your doctor. Be vigilant for signs and symptoms of adverse reactions. Seek immediate medical attention if you experience difficulty swallowing or speaking, breathing difficulties, or muscle weakness after treatment. Adverse reactions may occur within hours after treatment or several weeks later.
This drug binds to receptors at the neuromuscular junction, enters the nerve endings, and inhibits the release of acetylcholine, thereby blocking neuromuscular transmission. When injected into muscles in therapeutic doses, it causes localized muscle paralysis through chemical denervation. When muscles are chemically denervated, they become weakened and can develop acetylcholine receptors outside the synapse. The regeneration of nerves and the resumption of nerve impulses to the muscles demonstrate that the 'loss of strength' is reversible.
1) Pharmacological Action
This drug binds to receptors at the neuromuscular junction, enters the nerve endings, and inhibits the release of acetylcholine, thereby blocking neuromuscular transmission.
2) Clinical Trial Information
〈Lateral Canthal Lines〉
(A Phase 3 Clinical Trial) A double-blind, randomized, multi-center clinical trial was conducted with 204 adult patients (men and women aged between 18 and 75) requiring improvement of moderate to severe lateral canthal lines.
Participants were randomly assigned to receive either this drug or another botulinum toxin product, administered to either the right or left lateral canthal lines. Results showed that 4 weeks post-administration, the improvement rate of lateral canthal lines when smiling maximally was 65.02% (132 out of 203 participants) for the area treated with this drug, compared to 62.56% (127 out of 203 participants) for the area treated with the other botulinum toxin product. The lower limit of the 95% confidence interval for the difference in improvement rates of lateral canthal lines between the two treatment sites was -0.91%, indicating that the efficacy of this drug was not inferior compared to other botulinum toxin products. (Extended Clinical Trial) A single-group, open-label, multi-center extension clinical trial was conducted with 109 adults requiring improvement of moderate to severe lateral canthal lines and glabellar lines, who had participated in the aforementioned Phase 3 clinical trial.
Each participant received a single injection of 24 U into the lateral canthal lines and 20 U into the glabellar lines. As a result, the improvement rate of lateral canthal lines when smiling maximally 4 weeks after administration, as assessed by the investigators, was similar to the results of the Phase 3 clinical trial.